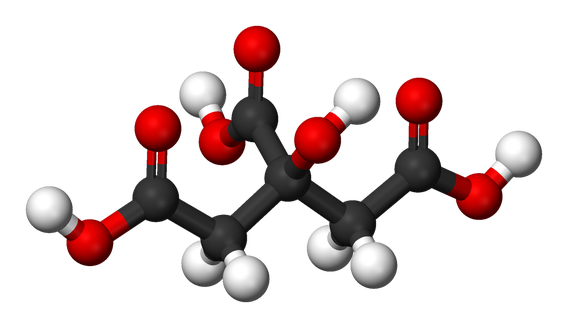
Citric Acid:
C6H6O7
Molecular Weight: 192.12352
Melting Point: 307.4 Fahrenheit
Boiling Point: According to the University of Wisconsin-Madison,
the boiling point of citric acid and its decomposition temperature is the same.
This means that when citric acid is heated above 175 degrees Celsius, the water
and carbon dioxide molecules separate from the acid. So it does not boil, before
it decomposes. (eHow)
Specific Gravity/Density: 1.665
g/cm3
pH: 1.7 (10% soln)
Physical State: Crystalline powder
Odor: Citric acid is
odorless
Appearance: White powder
C6H6O7
Molecular Weight: 192.12352
Melting Point: 307.4 Fahrenheit
Boiling Point: According to the University of Wisconsin-Madison,
the boiling point of citric acid and its decomposition temperature is the same.
This means that when citric acid is heated above 175 degrees Celsius, the water
and carbon dioxide molecules separate from the acid. So it does not boil, before
it decomposes. (eHow)
Specific Gravity/Density: 1.665
g/cm3
pH: 1.7 (10% soln)
Physical State: Crystalline powder
Odor: Citric acid is
odorless
Appearance: White powder
History of Citric Acid:

http://www.thebestjuicerreviews.com/image-files/lemons.jpg
The man who is credited for the discovery of citric acid was an
alchemist named Ibn Hayyan, but it has been known of since medieval times
through fruits (lemons and limes). It was not isolated until 1784 by a chemist
named Carl Wilhelm Scheel, who crystalized citric acid. It was a dormant
discovery until the 1860's when Italian citrus fruit industry rapidly
increased. Italian Citrus fruit was a major export until World War I when the
Italian export was disrupted. In 1893 a chemist named C. Wehmer found that
penacillium mold could extract the citric acid from sugar. This was called
microbial production of citric acid. This form of extraction was not used
indoustriously untill 1917 when an American chemist James Currie found that the
popular form of penicillium, Aspergillus niger, was an excellent producer of
citric acid. This technique was not used until two years later in large
quantities of production. (SolarNavigator.net)
alchemist named Ibn Hayyan, but it has been known of since medieval times
through fruits (lemons and limes). It was not isolated until 1784 by a chemist
named Carl Wilhelm Scheel, who crystalized citric acid. It was a dormant
discovery until the 1860's when Italian citrus fruit industry rapidly
increased. Italian Citrus fruit was a major export until World War I when the
Italian export was disrupted. In 1893 a chemist named C. Wehmer found that
penacillium mold could extract the citric acid from sugar. This was called
microbial production of citric acid. This form of extraction was not used
indoustriously untill 1917 when an American chemist James Currie found that the
popular form of penicillium, Aspergillus niger, was an excellent producer of
citric acid. This technique was not used until two years later in large
quantities of production. (SolarNavigator.net)